PHYLOGENETIC CLASSIFICATION
OF THE HEDWIGIACEAE
Contents of this website
|
||
| Phylogenetic classification |
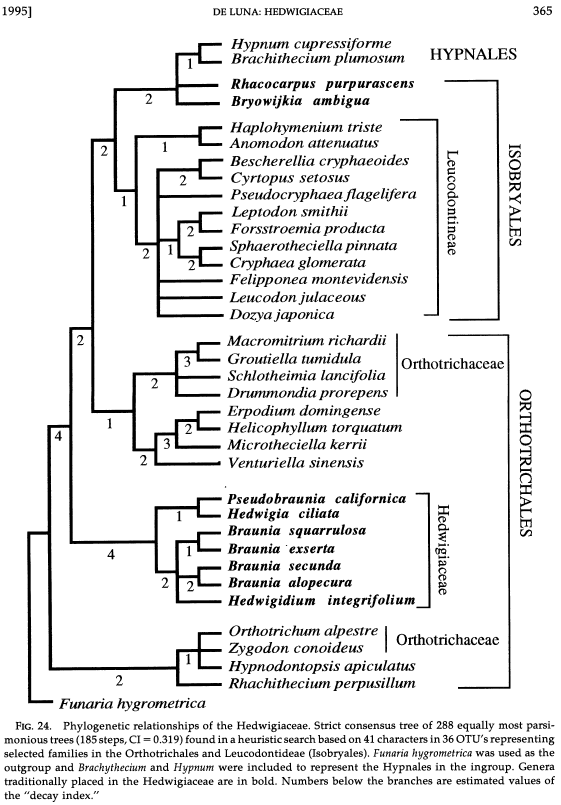
Ordinal relationships. The relationships and classification of the Hedwigiaceae were difficult to asses because morphological characters informative at the Order level are absent or were missinterpreted. Features of the peristome are missing in the family. Moreover, the gametophyte branching system was wrongly considered as being pleurocarpous. It is now clear that the plant habit in the Hedwigiaceae is acrocarpous. The archegonia are terminal on the main shoot, and the branching pattern is sympodial. The main axis of each plant consists of a succession of subterminal innovations, rather than a single shoot of indeterminate growth. The plants are plagiotropic and are pleurocarpous only in appearance (De Luna, 1990). |
|
The Hedwigiaceae was sometimes placed in the Grimmiales, Orthotrichales, or Leucodontales. Currently, the Hedwigiaceae is best placed in the Order Hedwigiales, likely sister to Order Bartramiales, among the acrocarpous mosses. Three families are grouped in the Order Hedwigiales. The Rhacocarpaceae is sister to the Hedwigiaceae, and the Helicophyllaceae is sister to both families. BRYOPHYTA Schimp. SUBDIVISION BRYOPHYTINA Engler CLASS BRYOPSIDA Rothm. SUBCLASS BRYIDAE Engl. SUPERORDER BRYANAE (Engl.) Goffinet & W. R. Buck
SUPERORDER HYPNANAE W. R. Buck, Goffinet & A. J. Shaw |
Summary of current inferences on the phylogenetic relationships of the Hedwigiaceae, based on De Luna (1995), Cox et al (2010), Chang and Graham (2014), and Ignatova et al (2016). |
The best phylogenetic hypothesis placing the Hedwigiaceae in the context of all mosses is the tree from Cox et al (2010) based on the maximum likelihood analyses of several molecular markers.
|
From: Cox CJ, et al. 2010. Moss diversity: A molecular phylogenetic analysis of genera. Phytotaxa 9: 175-195. |
The phylogenetic placement of the Hedwigiales among the orders of acrocarpous mosses seems to be sister to the Bartramiales, at the base of the Bryidae (Cox et al 2010). A moss phylogeny by Chang & Graham (2014) also revealed a relationship of the Hedwigiales at the base of the Bryidae.
|
|
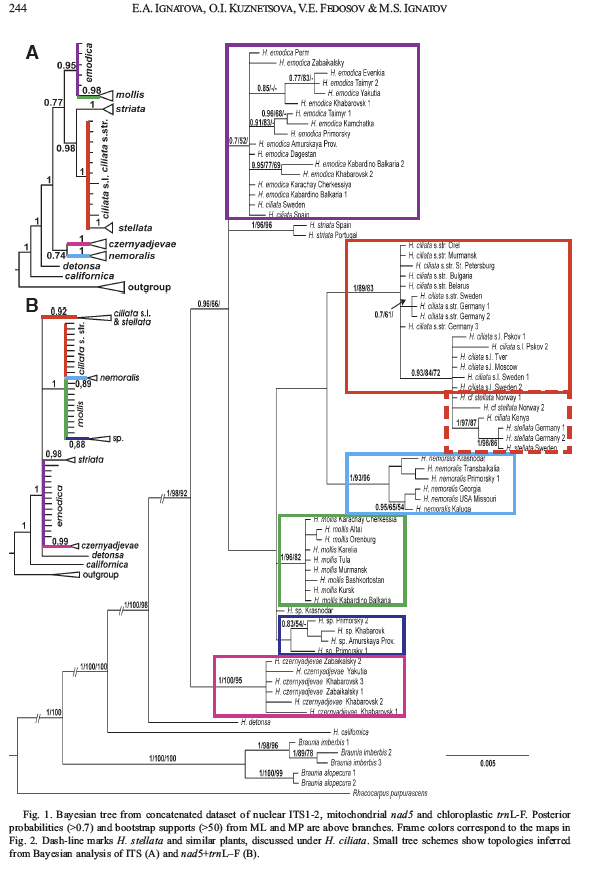
Relationships within the Hedwigiaceae. The Hedwigiaceae consists of four lineages ranked at generic level: Hedwigia, Pseudobraunia, Hedwigidium, and Braunia. The current circumscription of the Hedwigiaceae with four genera is based on morphological (De Luna 1995) and molecular phylogenetic analyses (Cox et al 2010). Bryowijkia and Rhacocarpus were sometimes included in the Hedwigiaceae. A phylogenetic analysis of morphological data suggested to exclude the two genera from the family (De Luna, 1995). According to Cox et al (2010) Bryowijkia is nested in the Dicranidae, sister to a clade formed by Aongstroemia, Garkea, Cladophascum and Eccremidium. The same analysis by Cox et al (2010) places Rhacocarpus as sister to the Hedwigiaceae. Within the Hedwigiaceae, Hedwigia and Pseudobraunia are sister taxa (De Luna 1995; Cox et al 2010), although Pseudobraunia californica is sometimes ranked as species in Hedwigia (Buchbender et al 2014; Ignatova et al 2016). Similarly, Braunia and Hedwigidium are sister genera (De Luna 1995, 2021; Cox et al 2010), although Hedwigidium imberbe is sometimes ranked as species in Braunia (Buchbender et al 2014; Ignatova et al 2016).
|
|
The molecular phylogenies by Buchbender et al (2014) investigated the relationships of several species in Hedwigia for the first time. They corroborated Pseudobraunia is related to Hedwigia, but found different relationships for Rhacocarpus: their likelihood analyses placed Rhacocarpus sister to Braunia. |
 |
A recent phylogeny of species of Hedwigia by Ignatova et al (2016) suggests phylogenetic relationships congruent with those found in De Luna (1995) and Cox et al (2010). One clade includes Braunia and Hedwigidium (as Braunia imberbis); this is sister to a clade which includes Pseudobraunia (as Hedwigia californica) and Hedwigia (Ignatova et al 2016). Rhacocarpus was considered outgroup to the clade including the four genera of Hedwigiaceae. These phylogenetic relationships grouping four taxa in the Hedwigiaceae are congruent with the distribution of synapomorphic morphological characters (globular protonema and papillose pseudoparaphylia) recorded for the four genera (De Luna 1995).
|
|
SYNAPOMORPHIES FOR THE HEDWIGIACEAE
|
|
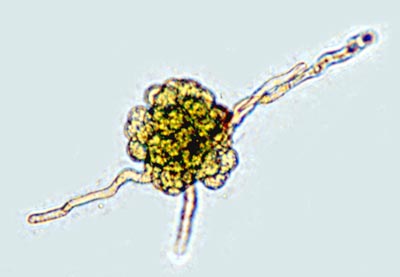
Globular protonema in the Hedwigiaceae |
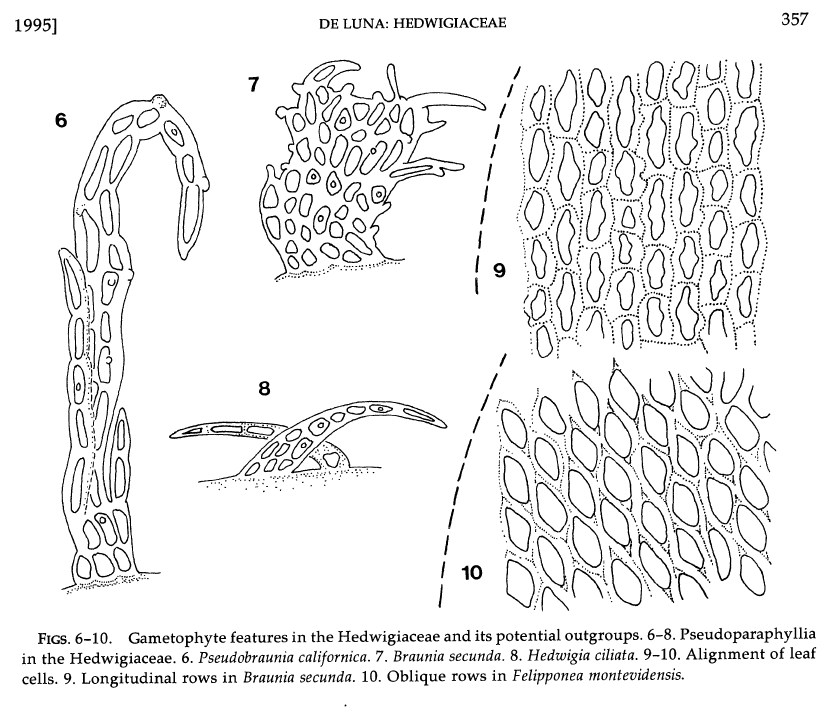 |
A combined morphological and molecular phylogenetic analysis of the Hedwigiaceae is still missing. Morphological synapomorphies for the Hedwigiaceae include
To date, these two characters have not been observed in Rhacocarpus, Bryowijkia, or any other moss. The protonema in Bryowijkia is endosporic, and the protonema in Rhacocarpus is filamentous, as is the majority of mosses (De Luna 1992). |
Fig. 6-8, papillose pseudoparaphylia ine the Hedwigiaceae (from De Luna 1995)
|
|
|
References Buchbender V, H. Hespanhol, M. Krug, C. Sérgio, A. Séneca, K. Maul, L. Hedenás & D. Quandt. 2014. Phylogenetic reconstructions of the Hedwigiaceae reveal cryptic speciation and hybridisation in Hedwigia. Bryophyte Diversity and Evolution 36(1): 1-21. Chang Y. & S. W. Graham. 2014. Patterns of clade support across the major lineages of moss phylogeny. Cladistics 30: 590-606. Cox C.J., B. Goffinet, A. J. Shaw, N.J. Wickett & S. B. Boles. 2010. Moss diversity: A molecular phylogenetic analysis of genera. Phytotaxa 9: 175–195. De Luna E. 1990. Protonemal development in the Hedwigiaceae (Musci), and its systematic significance. Systematic Botany 15: 192-204. De Luna E. 1990. Developmental evidence of acrocarpy in Hedwigia ciliata (Musci: Hedwigiaceae). Tropical Bryology 2: 53 60. De Luna E. 1992. Multicellular spores and false anisospory in Bryowijkia ambigua (Musci: Trachypodaceae). LINDBERGIA 16: 73-79. De Luna E. 1995. The circumscription and phylogenetic relationships of the Hedwigiaceae (Musci). Systematic Botany 20: 347-373. Ignatova EA, OI Kuznetsova, VE Fedosov & MS Ignatov. 2016. On the genus Hedwigia (Hedwigiaceae, Bryophyta) in Russia. Arctoa 25: 241-277. De Luna, E. 2021. Seta length variation and the refutation of Hedwigidium = Braunia (Hedwigiaceae, Bryopsida). Acta Botanica Mexicana. 128: e1810. DOI: https://doi.org/10.21829/abm128.2021.1810. Goffinet, B., W.R Buck and A.J. Shaw (2008), Morphology and classification of the Bryophyta, in Goffinet & Shaw (eds.) Bryophyte Biology 2nd edition, p. 55–138, Cambridge University Press. |
|
FROM THE ONLINE VERSION OF FLORA OF NEW ZEALAND. Based on Fife, AJ. 2015 Hedwigiaceae. Taxonomy
The relationships and circumscription of the Hedwigiaceae have been,
and remain, controversial. The Hedwigiaceae were placed in the order
Isobryales by Brotherus (1925). He subdivided the family into 3 subfamilies with the Hedwigioideae the largest, with 4 genera. De Luna (1995) re-evaluated the limits of the family and its relationships; he considered the 4 genera (Hedwigia, Hedwigidium, Braunia, and Pseudobraunia)
placed by Brotherus in the Hedwigioideae to comprise the monophyletic
family Hedwigiaceae, which he considered supported by 2 unique
synapomorphies: papillose pseudoparaphyllia and globular protonema. De
Luna’s circumscription of the family agrees with that subsequently
adopted by Goffinet et al. (2009), who placed the family in the order Hedwigiales.
Hedwigia and Hedwigidium are small genera with representatives in New Zealand. Braunia is a tropical genus of c. 23 species occurring on rock in the Americas, Africa, and Asia. Pseudobraunia is a monotypic genus confined to western North America. A fifth genus, Bryowijkia (=Cleistostoma), of eastern Asia, is also sometimes placed in this family. The distinctive globular protonemata, mainly composed of rounded cells, were illustrated by de Luna (1990), using cultured material. While the genus Rhacocarpus was placed in the Hedwigiaceae by Brotherus (1925), there is an increasing consensus that it deserves recognition at the family level, in large part because of its distinctive laminal cell ornamentation (see Barthlott & Schultze-Motel 1981). According to Crum (1994, p. 667) Rhacocarpus resembles the Hedwigiaceae “only in lacking a costa and a peristome”. De Luna (1990) demonstrated that the protonema of Rhacocarpus was not globular. The removal of the Rhacocarpaceae from the Hedwigiaceae was advocated by de Luna (1995) and followed by Goffinet et al. (2009).
References:
Barthlott, W.; Schultze-Motel, W. 1981: Zur Feinstruktur der Blattoberflächen und systematischen Stellung der Laubmoosgattung Rhacocarpus and anderer Hedwigiaceae. Willdenowia 11: 3–11.
Brotherus, V.F. 1925: Musci (Laubmoose). In: Engler, A. (ed.) Die natürlichen Pflanzenfamilien. Edition 2. Bd 11. Engelmann, Leipzig. 1–542.
Crum, H.A. 1994: Rhacocarpaceae. In: Sharp, A.J.; Crum, H.A.; Eckel, P.M. (ed). The Moss Flora of Mexico. Memoirs of the New York Botanical Garden 69: 667.
de Luna, E. 1990: Protonemal development in the Hedwigiaceae (Musci) and its systematic significance. Systematic Botany 15: 192–204.
de Luna, E. 1995: The circumscription and phylogenetic relationships of the Hedwigiaceae (Musci). Systematic Botany 20: 347–373.
Fife, A.J. 2014: Hedwigiaceae. In: Heenan, P.B.; Breitwieser, I.; Wilton, A.D. Flora of New Zealand — Mosses. Fascicle 14. Manaaki Whenua Press, Lincoln.
Goffinet, B.; Buck, W.R.; Shaw, A.J. 2009: Morphology, anatomy, and classification of the Bryophyta. In: Goffinet, B.; Shaw, A.J. (ed.) Bryophyte Biology. Edition 2. Cambridge University Press, Cambridge. 55–138.
|
FROM THE ONLINE VERSION OF CENTRAL AMERICAN MOSSES Hedwigiaceae. Discussion The Hedwigiaceae consist of four, very similar genera: two with exserted capsules (Braunia Bruch & Schimp. in B.S.G., Pseudobraunia (Lesq. & James) Broth.), and two with immersed ones (Hedwigia P. Beauv., Hedwigidium Bruch & Schimp. in B.S.G.). The gametophytes of Hedwigia and Pseudobraunia are so similar they are sometimes considered synonymous (De Luna 1992) despite their different setae lengths. Hedwigia and Hedwigidium have also been synonymized (Nyholm 1960;
|
| |

 The branching system in Hedwigia consists of a series of sympodial modules
The branching system in Hedwigia consists of a series of sympodial modules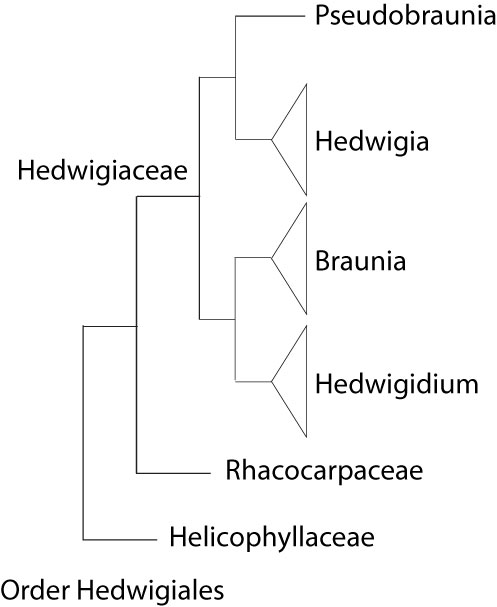
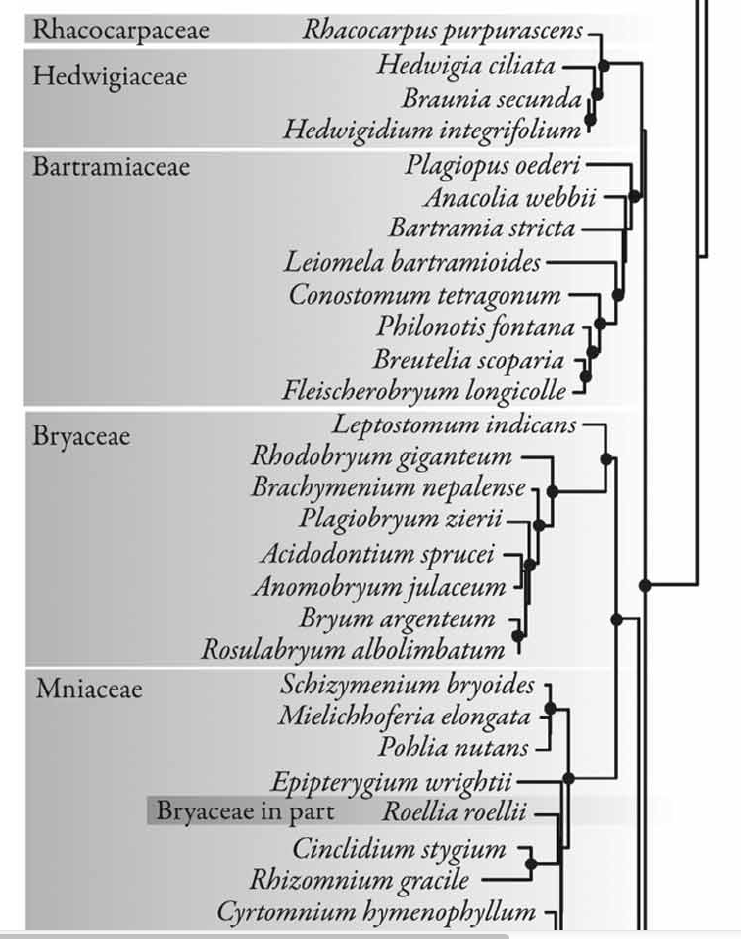
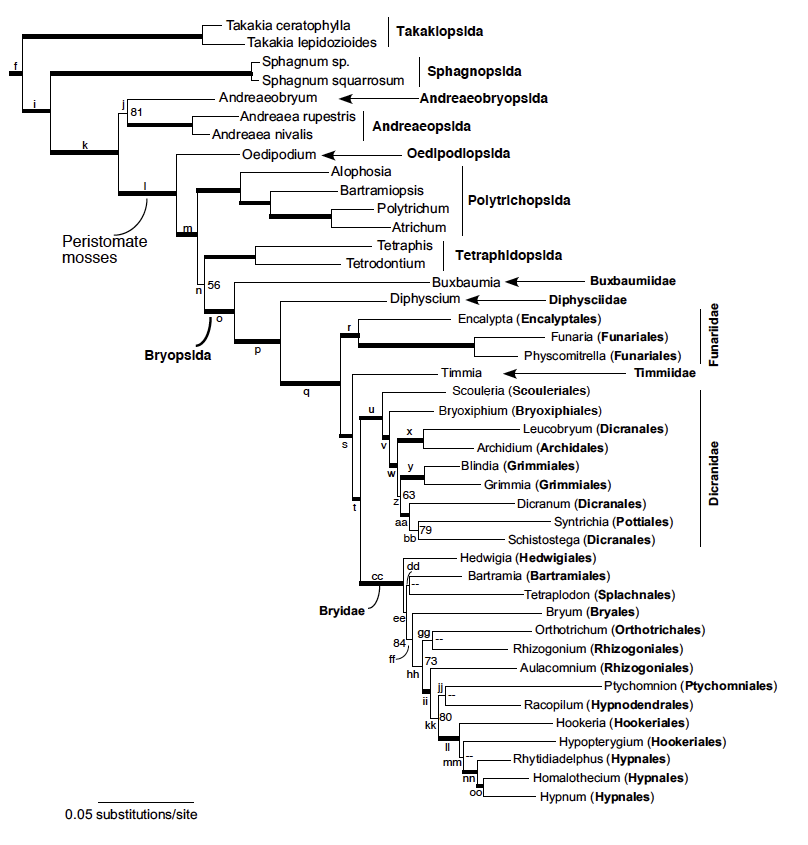 Fig 1. Moss phylogeny inferred in an unpartitioned likelihood analysis of plastid data with base frequencies optimized (using Garli). Bootstrap values for parsimony and likelihood analysis (the latter an unpartitioned Garli analysis with optimized base frequencies), respectively, are indicated beside branches; those with < 50% support are indicated with a dash (–), and those with > 90% bootstrap support in both analyses are indicated with thick lines. Names of major taxa are indicated, including orders in subclasses Bryidae, Dicranidae, and Funariidae. Clades along the phylogeny referred to in the text are indicated with labels; see Table 2b for bootstrap support in other analyses. This is a subset of a larger green plant tree, shown in Fig. S2. The scale bar indicates estimated substitutions per site. (from Chang and Graham, 2014)
Fig 1. Moss phylogeny inferred in an unpartitioned likelihood analysis of plastid data with base frequencies optimized (using Garli). Bootstrap values for parsimony and likelihood analysis (the latter an unpartitioned Garli analysis with optimized base frequencies), respectively, are indicated beside branches; those with < 50% support are indicated with a dash (–), and those with > 90% bootstrap support in both analyses are indicated with thick lines. Names of major taxa are indicated, including orders in subclasses Bryidae, Dicranidae, and Funariidae. Clades along the phylogeny referred to in the text are indicated with labels; see Table 2b for bootstrap support in other analyses. This is a subset of a larger green plant tree, shown in Fig. S2. The scale bar indicates estimated substitutions per site. (from Chang and Graham, 2014)